Question: What is the optimum temperature for the
Geobacillus stearothermophilus amylase?
Hypothesis: The optimum temperature for the amylase obtained from
Geobacillus stearothermophilus is high, most probably above 60 ℃ since the bacteria mainly lives in high temperature environments such as hot springs.
If the temperature of the reaction environment rises above 60 ℃, then the amount of maltose formed from the reaction of the bacterial amylase, will be the highest concentration.
Reagents: Distilled water, Buffer
, 5% starch solution, bacterial amylase, 3,5-Dinitrosalicylic acid (DNS)
Equipment: 12 test tubes, test tube holder, water bath, spectrophotometer, cuvettes, micropipettes.
Procedure
For the determination of the effect of temperature on the activity of
Geobacillus stearothermophilus amylase, 6 test tubes are labelled and placed in a test tube rack.
450 μl of distilled water is added to each test tube followed by the same amount of buffer solution that helps to maintain the pH of the reaction environment.
450 μl of 5% starch is them added to the six test tubes, followed by 100 μl of the bacterial enzyme. The tubes containing the mixture are then placed in water bath calibrated at varying temperatures (0℃, 25℃, 37℃, 45℃, 65℃, and 85℃) for 10 minutes.
Afterwards the tubes are recovered from the boiling water bath and 1 ml of DNS is added before being transferred to a boiling water bath for 5 minutes. After the 5 minutes have elapsed, the tubes are recovered, and 8 ml is added to dilute the contents.
The absorbance of each tube is then determined by transferring part of the solution to a cuvette and then taking readings using the spectrophotometer. The contents of the first tube can be used as blank solution to reset the spectrophotometer.
The absorbance of solution from each test tube corresponds to the amount of maltose produced at each given temperature, which also reflect the level of the amylase activity.
DATA ANALYSIS
Experiment setup for temperature
| Tube |
Water (μl) |
5% Starch (μl) |
Amylase (μl) |
Temperature (0C) |
| 1 |
450 |
450 |
100 |
0 |
| 2 |
450 |
450 |
100 |
25 |
| 3 |
450 |
450 |
100 |
37 |
| 4 |
450 |
450 |
100 |
45 |
| 5 |
450 |
450 |
100 |
65 |
| 6 |
450 |
450 |
100 |
85 |
Data from the study of the Effect of Temperature on Bacterial Amylase Activity
| Temperature (℃) |
Absorbance at 540nm |
Maltose produced (mg/ml) |
Amylase Activity (mg/ml/min) |
| 0 |
0.116 |
0.026 |
0.0026 |
| 25 |
0.237 |
0.049 |
0.0049 |
| 37 |
0.309 |
0.063 |
0.0063 |
| 45 |
0.391 |
0.079 |
0.0079 |
| 65 |
0.895 |
0.176 |
0.0177 |
| 85 |
1.029 |
0.201 |
0.0202 |
Concentration of maltose produced was read directly on the standard curve in part 2
Formula used to calculate the activity of maltose: [Maltose (mg/ml) ÷ Time (min)]
For instance: Maltose produced in tube 2: 0.049
Time incubated: 10 minutes
Therefore activity: (0.049 ÷10) = 0.0049 mg/ml/min
Graph of bacterial amylase activity vs. temperature:
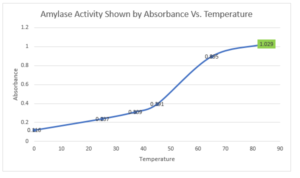
The optimum temperature is shown in green color.
The standard curve in part II shows that the absorbance of amylase increases with increasing concentration. The second graph showing on the optimum activity of amylase from
Geobacillus stearothermophilus shows that the amount of maltose produced increases with increasing temperature. At around 45℃, the amount of products increases sharply before slowing down from around 70℃ and flattening at 85℃. The amount of maltose produced corresponds to enzyme activity, which means that the optimum temperature for
Geobaciollus thermophilus’ amylase is high at around 85℃.
Conclusion
My hypothesis was that the amylase from
Geobacillus stearothermophilus has an optimum. temperature of about 60 ℃. According to Kotzekidou (1999), the bacteria from which the enzyme has been obtained is a “thermophilic, aerobic, spore-forming bacterium,” that grows maximally at a temperature of 65-75℃. Based on the results from the standard curve I reject my previous hypothesis, since the findings have shown that the amylase has an optimum temperature of about 85℃. Amylase is an important enzyme that enables
Geobacillus stearothermophilus to metabolize starch in harsh environments such as hot springs. Possible sources of errors in this experiment include improper calibration of the reaction time and temperature. Further experiments could be used to determine the optimum temperature for enzymes obtained from mesophiles or psychrophiles.
Reference
Kotzekidou, P. (2014). Geobacillus stearothermophilus (Formerly Bacillus stearothermophilus).
Encyclopedia of Food Microbiology, 129-134.
Jain, A., Jain, R., & Jain, S. (2020). Quantitative analysis of reducing sugars by 3, 5-dinitrosalicylic acid (DNSA method). In
Basic Techniques in Biochemistry, Microbiology and Molecular Biology (pp. 181-183). Humana, New York, NY.
 The optimum temperature is shown in green color.
The standard curve in part II shows that the absorbance of amylase increases with increasing concentration. The second graph showing on the optimum activity of amylase from Geobacillus stearothermophilus shows that the amount of maltose produced increases with increasing temperature. At around 45℃, the amount of products increases sharply before slowing down from around 70℃ and flattening at 85℃. The amount of maltose produced corresponds to enzyme activity, which means that the optimum temperature for Geobaciollus thermophilus’ amylase is high at around 85℃.
Conclusion
My hypothesis was that the amylase from Geobacillus stearothermophilus has an optimum. temperature of about 60 ℃. According to Kotzekidou (1999), the bacteria from which the enzyme has been obtained is a “thermophilic, aerobic, spore-forming bacterium,” that grows maximally at a temperature of 65-75℃. Based on the results from the standard curve I reject my previous hypothesis, since the findings have shown that the amylase has an optimum temperature of about 85℃. Amylase is an important enzyme that enables Geobacillus stearothermophilus to metabolize starch in harsh environments such as hot springs. Possible sources of errors in this experiment include improper calibration of the reaction time and temperature. Further experiments could be used to determine the optimum temperature for enzymes obtained from mesophiles or psychrophiles.
Reference
Kotzekidou, P. (2014). Geobacillus stearothermophilus (Formerly Bacillus stearothermophilus). Encyclopedia of Food Microbiology, 129-134.
Jain, A., Jain, R., & Jain, S. (2020). Quantitative analysis of reducing sugars by 3, 5-dinitrosalicylic acid (DNSA method). In Basic Techniques in Biochemistry, Microbiology and Molecular Biology (pp. 181-183). Humana, New York, NY.
The optimum temperature is shown in green color.
The standard curve in part II shows that the absorbance of amylase increases with increasing concentration. The second graph showing on the optimum activity of amylase from Geobacillus stearothermophilus shows that the amount of maltose produced increases with increasing temperature. At around 45℃, the amount of products increases sharply before slowing down from around 70℃ and flattening at 85℃. The amount of maltose produced corresponds to enzyme activity, which means that the optimum temperature for Geobaciollus thermophilus’ amylase is high at around 85℃.
Conclusion
My hypothesis was that the amylase from Geobacillus stearothermophilus has an optimum. temperature of about 60 ℃. According to Kotzekidou (1999), the bacteria from which the enzyme has been obtained is a “thermophilic, aerobic, spore-forming bacterium,” that grows maximally at a temperature of 65-75℃. Based on the results from the standard curve I reject my previous hypothesis, since the findings have shown that the amylase has an optimum temperature of about 85℃. Amylase is an important enzyme that enables Geobacillus stearothermophilus to metabolize starch in harsh environments such as hot springs. Possible sources of errors in this experiment include improper calibration of the reaction time and temperature. Further experiments could be used to determine the optimum temperature for enzymes obtained from mesophiles or psychrophiles.
Reference
Kotzekidou, P. (2014). Geobacillus stearothermophilus (Formerly Bacillus stearothermophilus). Encyclopedia of Food Microbiology, 129-134.
Jain, A., Jain, R., & Jain, S. (2020). Quantitative analysis of reducing sugars by 3, 5-dinitrosalicylic acid (DNSA method). In Basic Techniques in Biochemistry, Microbiology and Molecular Biology (pp. 181-183). Humana, New York, NY.

